 The nuclear binding energy varies between nuclei.This increases the mass of nuclei with more neutrons than protons relative to the atomic mass unit scale based on 12C with equal numbers of protons and neutrons. The neutron is slightly heavier than the proton.There are two reasons for the difference between mass number and isotopic mass, known as the mass defect: For example, 63Cu (29 protons and 34 neutrons) has a mass number of 63, and an isotopic mass in its nuclear ground state is 62.91367 u. The isotopic mass usually differs for other isotopes and is usually within 0.1 u of the mass number. One unified atomic mass unit is approximately the mass of one nucleon (either a single proton or neutron) and is numerically equivalent to 1 g/mol.įor 12C, the atomic mass is exactly 12u since the atomic mass unit is defined from it. One atomic mass unit is equal to 1.66 x 10 -24 grams. The unit of measure for mass is the atomic mass unit (amu). 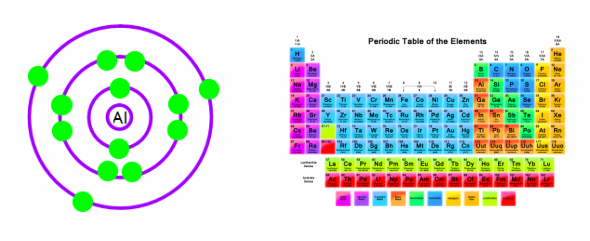
Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally-occuring isotopes and their abundance. See also: Properties of Aluminum Atomic Mass of Aluminum 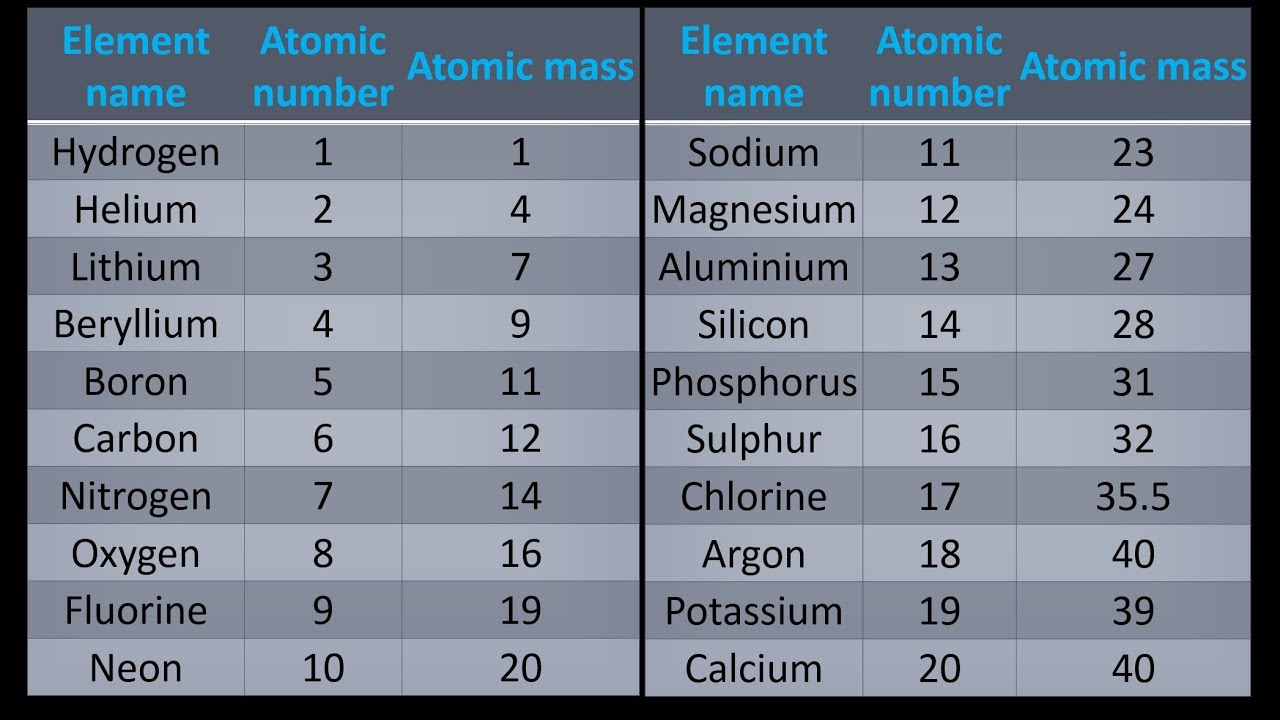 
By mass, aluminium makes up about 8% of the Earth’s crust it is the third most abundant element after oxygen and silicon and the most abundant metal in the crust, though it is less common in the mantle below.Īluminum – Properties Element Aluminum Atomic Number 13 Symbol Al Element Category Poor Metal Phase at STP Solid Atomic Mass 26.9815 Density at STP 2.7 Electron Configuration 3s2 3p1 Possible Oxidation States +3 Electron Affinity 42.5 Electronegativity 1.61 1st Ionization Energy 5.9858 Year of Discovery 1825 Discoverer Oersted, Hans Christian Thermal properties Melting Point 660 Boiling Point 2467 Thermal Conductivity 237 Specific Heat 0.9 Heat of Fusion 10.79 Heat of Vaporization 293.4 The chemical symbol for Aluminum is Al.Īluminium is a silvery-white, soft, nonmagnetic, ductile metal in the boron group. Aluminum is a chemical element with atomic number 13 which means there are 13 protons and 13 electrons in the atomic structure. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
